The advent of Artificial Intelligence (AI) has ushered in a new era of technological innovation, offering unprecedented opportunities for businesses to enhance efficiency, innovation, and competitiveness. However, the journey to becoming AI-ready involves navigating a complex landscape of technical, organizational, and cultural challenges. The guide below and the subsequent checklist provide actionable advice for data leaders and practitioners on preparing their organizations to incorporate AI.
Technical Preparation:
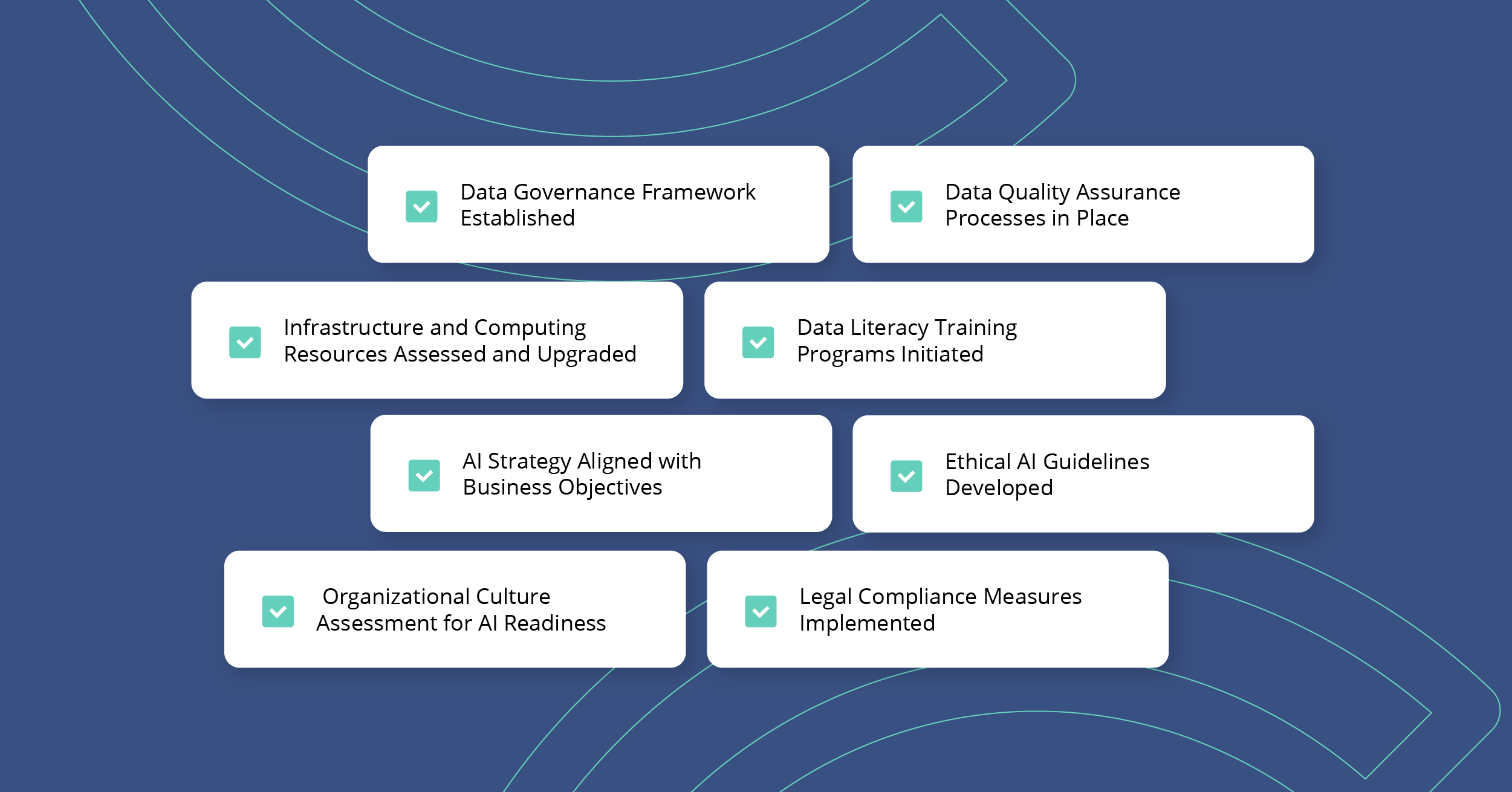
Enhance Data Accessibility and Quality
- Establish a robust data governance framework to ensure data accuracy, consistency, and security.
- Implement data cleaning and preprocessing techniques to improve data quality.
- Foster partnerships with data providers to access diverse and extensive datasets.
Upgrade Infrastructure and Computing Resources:
- Invest in scalable cloud computing services to handle large-scale AI computations.
- Conduct regular technology audits to identify and address infrastructure gaps.
- Explore high-performance computing solutions tailored to AI applications.
Boost Data Literacy Across the Organization
- Assess current skill levels and identify gaps in data literacy.
- Develop comprehensive training programs focusing on data analysis, interpretation, and AI applications.
- Encourage continuous learning and upskilling in AI and data science.
Organizational Changes
Cultivate an AI-Ready Culture
- Promote a culture of innovation and openness to new technologies.
- Educate stakeholders on the benefits and potential of AI.
- Encourage cross-departmental collaboration to integrate AI insights into various business processes.
Address Ethical and Legal Concerns
- Develop ethical guidelines for AI usage, focusing on fairness, accountability, and transparency.
- Stay informed about legal regulations related to AI and data privacy.
- Implement compliance measures and regular audits to ensure ethical AI practices.
Strategic AI Implementation Planning
- Develop a clear AI strategy aligned with business objectives.
- Identify key areas where AI can add the most value.
- Establish measurable goals and KPIs to track AI implementation success.
Cultural Adaptation
Overcome Resistance to Change
- Communicate the vision and benefits of AI clearly to all levels of the organization.
- Involve employees in the AI integration process and decision-making.
- Recognize and address concerns and fears related to AI adoption.
Foster a Collaborative Environment
- Encourage open communication and knowledge sharing about AI projects.
- Create multidisciplinary teams to bring diverse perspectives to AI initiatives.
- Celebrate successes and learn from setbacks in AI projects.